The use of 3D printing in the medical sector reveals that the technology is already exploited in 7 fields of application: orthopedic surgery, maxillofacial surgery, reconstructive plastic surgery, ENT surgery, neurosurgery, cardiac surgery and urological surgery.
While the technology is increasingly being used in medical applications, its main benefits (time saving and pre-operative planning) are almost always highlighted compared to the real challenges encountered by players. Those challenges often include legal restrictions or communication barriers at the level of the process implementation.
A talk with 3D-Side sheds light on these issues in such vital area and reveals that, “just because [one] can print [a model], doesn’t mean [one] can use it”.
3D-Side, a company that delivers a patient specific surgical technology”
3D-Side is a medical technology company founded by Khanh Tran Duy and Laurent Paul. Mechanical engineer by training, Khanh Tran Duy completed a thesis in maxillofacial surgery assistance and Laurent Paul focused his research on surgical assistance for bone tumor. They decided to combine their respective expertise in the surgical field to additive manufacturing and software in order to create 3D-Side. Healthcare professionals can benefit from these services at the international level.
While taking advantage of the results of their thesis, 3D-Side aims to add value to the way surgeons work. Technically speaking, the 3D-Side team uses the patient’s medical images to elaborate a 3D planning that best describes the procedure that fits his/her need. Once the medical device has been produced, it is sent to the hospital in charge of the patient.
After more than 10 years in R&D, the team has launched “Customize”, a platform that facilitates the creation of patient specific medical devices on the one hand, on the other hand, ensures communication with surgeons and sets up a complete ISO13485 workflow to plan complex interventions.
The use of 3D Printing in the medical field
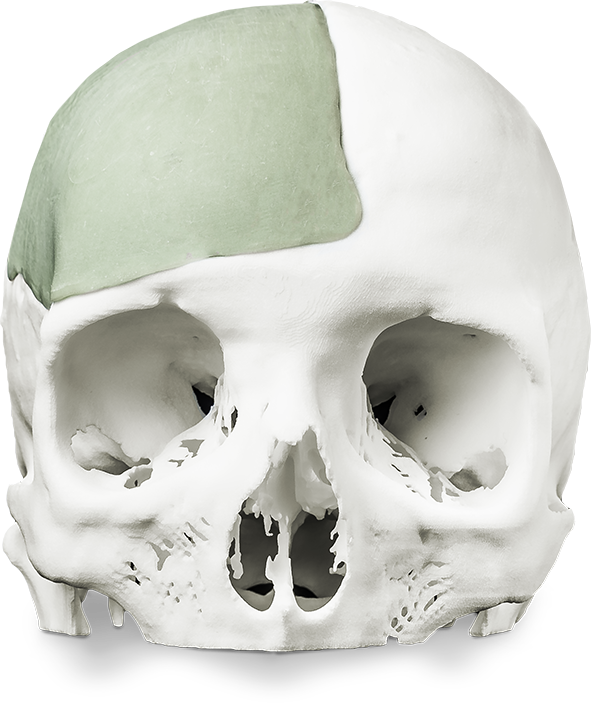
3D printing or additive manufacturing consists in making three dimensional solid objects from a digital file, using appropriate materials (metal, plastic, ceramic or resin). Until now, the manufacturing process seems pretty much easy to achieve once the user has a 3D printer and the required materials.
However, in the medical field, a number of elements make the use of a 3D printed model complex. That’s why Khanh Tran Duy mentions the difference between “making an STL. file” and “being the legal manufacturer of a medical device (MD)”: “In the healthcare industry, having a 3D printer, software and appropriate materials is not enough. The medical industry establishes a clear difference between the manufacturing process and the legal manufacturer. The legal manufacturer has to comply with the European directives on the use of 3D printing in the medical sector. He takes on responsibility of ensuring his/her technology is compatible as well as responsibility of each 3D printed model, which requires a number of validations to confirm the use of the 3D printed model and its performance.”
Legal aspects to consider regarding 3D printed medical devices
At the European Union level, 5 main aspects frame the use of 3D printing for the manufacturing of medical devices. Obviously, the application of these aspects may vary from one country to another according to the current legislation:
– Requirements for market access. In general, Khanh Tran Duy explains that a series of actions have been implemented to improve traceability of 3D printed MD. This general requirement includes that distributors and importers must collaborate with manufacturers, so that every stakeholder can identify the previous and next participant in the supply chain. At the level of 3D-Side for instance, the platform “Customize” makes it possible to ensure collaboration between all involved parties in a project. A “co-design” module allows the surgeon to modify and validate the prototype before its production, to handle the production process and to ensure that the right person receives the right information at the right time.
– Liability. As the co-founder of 3D-Side states: “the producer is liable for any defect in its product. As far as 3D printing is concerned, there is a deviation from the traditional chain of production, distribution and use. Many parties are involved in the production of 3D devices in this case: the surgeon who makes the initial design, the software engineer who develops the 3D design, the 3D printer’s producer, the materials supplier, the implanting surgeon, the hospital, etc. The complete ISO13485 workflow must carefully be applied. Furthermore, professionals should keep in mind that new regulations will be effective as from May 2020. The EU-MDR (Medical Device Regulation) strongly strengthens all these applications.”
In addition to these two aspects, one notes 3 other legal aspects drawn from the KCE (Belgian Health Care Knowledge Centre) report on the use of high-risk medical devices: patient rights, protection of patient data and intellectual property rights.
Medical devices and mass personalisation

As in every area of application of 3D printing, mass production and/or mass customization are also part of the concerns raised. In the medical field, there are two types of “tailor-made” MD: patient specific MD and custom-made/personalized MD (CE Marking required).
According to Khanh, in contrast to patient specific MD, manufacturers of personalized medical devices medical devices, regardless of the risk profile, should apply a CE marking to their product. The patient specific one remains unmarked and requires a mandatory validation process (for each patient) as well as an appropriate PMS (post-marketing surveillance), which takes a lot of time. For these devices, “although it is complex, mass customization can be achieved with our software”, concludes Khanh.
Challenges and development opportunities
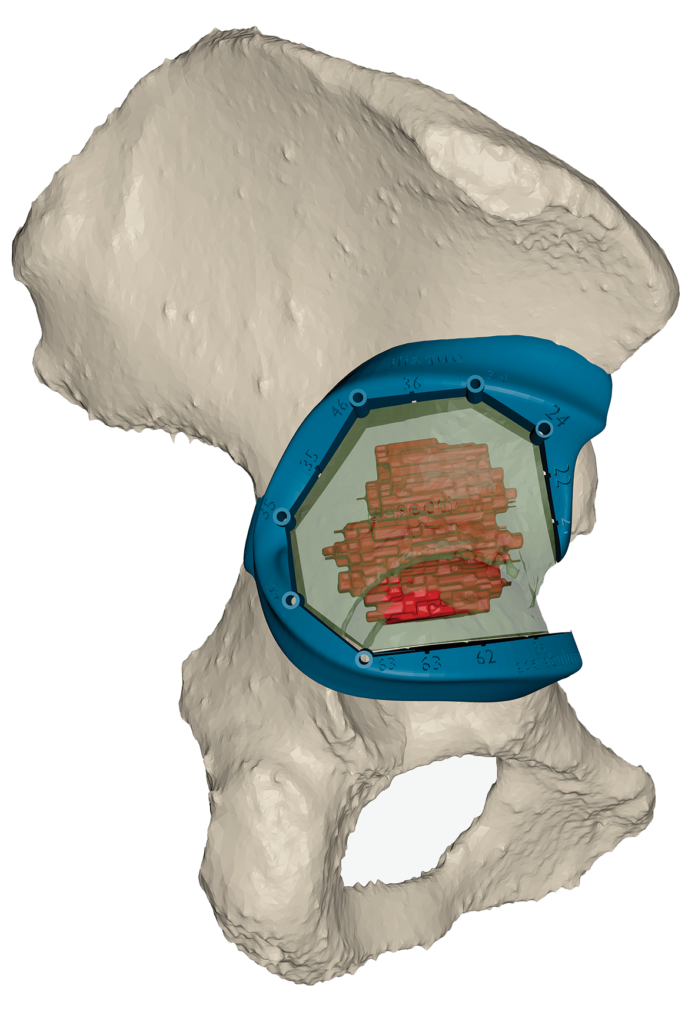
Speaking of challenges encountered by the use of 3D printing in the medical sector, the financial aspect remains a substantial hurdle to its full integration.
Indeed, despite the advantages of a 3D printed guide, the price of a 3D printed MD remains expensive and this is also due to the product liability insurance and required validations.
According to our resource person, in Europe, MD are not that expensive compared to other markets. However, European countries vary from one to another and apply the requirements for market access according to the existing law.
Surprisingly perhaps, hospitals cannot 3D print their own MD. They must be granted a certain number of qualifications first, not to mention, the mastering of new skills required by 3D printing technologies.
Companies keep developing 3D printing technologies, but they are in fact a “small” step in the production of a MD. The ratio cost/benefit must always be controlled and measured. For the patient and social security.
Despite these financial barriers, the co-founder of 3D-Side sees a future where 3D printing will enable mass customization and where professionals will feel more confident in the use of 3D printing technologies. Lastly, manufacturers, should always keep an eye on regulations, because “just because you can print it, doesn’t mean you can use it,” concludes Khanh Tran Duy.
This interview has initially been published in the 2019 April-May Issue of 3D ADEPT Mag.
Remember, you can post job opportunities in the AM Industry on 3D ADEPT Media free of charge or look for a job via our job board. Make sure to follow us on our social networks and subscribe to our weekly newsletter : Facebook, Twitter, LinkedIn & Instagram ! If you want to be featured in the next issue of our digital magazine or if you hear a story that needs to be heard, make sure you send it to contact@3dadept.com






