Insights from Materialise’s 3D Planning and Printing in Hospitals Forum, 8th Edition
Aaron James lost half his face after being electrocuted on the job. Nobody would wish that on anyone. When Koen Peters, EVP of Materialise’s Medical unit, opened the 8th edition of the 3D Printing in Hospitals Forum with Aaron’s story, the room went quiet, from recognition.
We covered Aaron’s landmark surgery at the time, a world first in which Materialise’s 3D-printed cutting instruments helped surgeons perform a full face and eye transplant. Today, Aaron is doing well. He has returned to a version of normalcy. He is living proof of what becomes possible when technology, planning, and surgical precision converge around a single patient’s anatomy.
It is one of these stories I like to call “medical miracles”. It is also, by Peters’s own admission, not a sustainable model for the future of healthcare.

“The value for the patients is almost obvious,” Koen Peters noted. “But the language that authorities and reimbursement boards speak is the language of numbers. That’s the job we really have to do: demonstrate the value in a very structured way, based on data.”
Don’t get me wrong: I am not saying that exceptional cases like Aaron’s don’t matter. In fact, they matter profoundly. But if there is one thing I have taken from my time at Materialise’ conference is that adoption of 3D technology in hospitals cannot be built on one-off successes.
It requires systems, workflows, reproducible results, and most importantly, daily planning that never makes the news.
A misconception that has shaped how much of the industry talks about 3D technology in healthcare
 There is a misconception that has shaped how much the industry talks about 3D technology in healthcare — and as a trade publication, we have certainly contributed to putting 3D printing on such a pedestal. The assumption is that the end goal is always 3D printing. It is not. Or at least, it is not always. And this forum made it clear.
There is a misconception that has shaped how much the industry talks about 3D technology in healthcare — and as a trade publication, we have certainly contributed to putting 3D printing on such a pedestal. The assumption is that the end goal is always 3D printing. It is not. Or at least, it is not always. And this forum made it clear.
Across specialties (cranio-maxillofacial surgery, oncology, cardiology, urology, orthopedics, and pediatrics) speaker after speaker described how 3D planning alone had changed what happened in the operating room. Because they were able to see the anatomy before making the first incision.
One figure from the forum stood out: 3D models changed the surgical plan in 44% of cases involving congenital heart disease. Nearly half. Which means that without the 3D model, nearly half of those surgical approaches would have been based on an incomplete picture.
Consider what that means in practice. In a complex oncological case (a mandible reconstruction using pieces of fibula, for instance,) planning the geometry of those bone segments in software can reduce a procedure that might otherwise take hours of improvisation to a 5–10-minute pre-mapped sequence.
As Arsham Makaryan, application engineer, at Materialise told me during a software demonstration: every minute saved in the operating room means saving the time of 10 to 15 people simultaneously. “All the planning, all the pre-planning that you can do before surgery saves those people’s time at the same time.”
What clinical applications are actually teaching us
The breadth of use cases presented at this year’s forum reflected just how far point-of-care 3D has spread beyond its original strongholds in CMF and orthopedics.
In cardiology, cardiologist Joan Sanchez-de-Toledo shared how 3D models of congenital heart disease, particularly complex pediatric anatomies, have transformed both surgical preparation and family communication. The same model that helps a surgeon understand where to cut helps a parent understand what their child is facing. The software, he noted, gives teams the opportunity to adjust to the patient: to the family, to the surgeon, to the specific anatomy.
In oncology and reconstructive surgery, the forum highlighted how virtual planning for mandibular reconstruction where surgeons piece together resected jaw sections using fibula bone segments has become feasible and efficient. What once required improvisation under pressure in the OR now has a roadmap.
In general and urological surgery, teams from institutions including the eDIMES Lab presented how preoperative planning and intraoperative navigation are being applied across vascular, gynecological, and urological procedures, areas where 3D planning is still gaining ground.
In CMF and orbital surgery, a point-of-care 3D lab shared how conventional planning had historically produced implant deviations of up to 40 degrees. With 3D-guided approaches and real-time intraoperative navigation, those margins have been dramatically reduced. The future they are building toward, they noted, includes AI, augmented reality, and robotics.
Mimics: the common thread across these applications
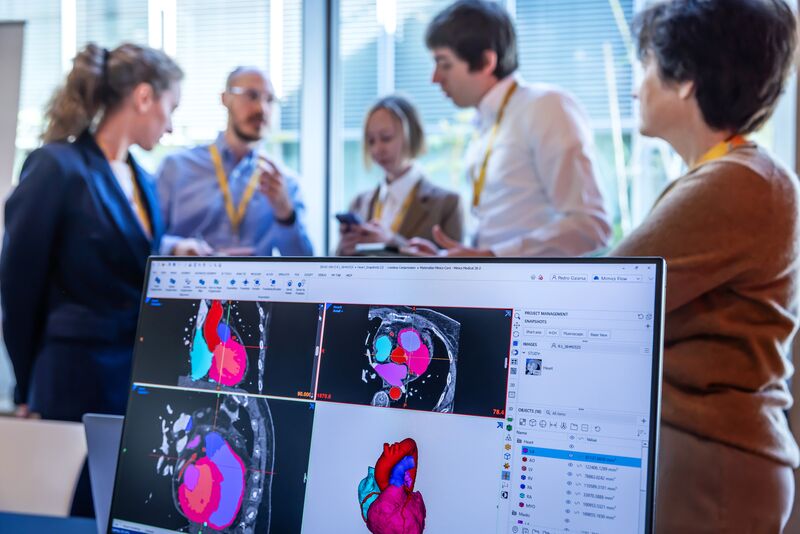
Running beneath nearly every clinical application presented at the forum is Materialise’s Mimics software platform, the infrastructure through which CT scans become navigable 3D anatomies, and anatomies become surgical plans.
Mimics is best understood as a software ecosystem. At its core is Mimics Core, the full engineering toolbox that allows users to segment CT and MRI data, generate surface models of any anatomical structure, and perform measurements, design surgical guides, or simulate procedures. It is used by biomedical engineers, clinical engineers, and an increasing number of surgeons directly.
The segmentation process, converting a CT scan’s stacked 2D slices into a manipulable 3D model, is more nuanced than it appears. Different tissues have different radiodensity values; selecting the right threshold determines whether you are visualizing a heart or a rib cage. As Materialise’s application engineer Arsham Makaryan demonstrated: “It’s up to me to pick the right threshold. That’s what it all boils down to.”
From that model, the software allows users to isolate, clean, color-code, measure and export any structure of interest.
Some segmentations, particularly those involving MRI data or soft tissue oncological cases, can take two hours or more. Others are completed in minutes. The software adapts to the case.
Alongside Mimics Core sits a portfolio of surgical-specific planners, including Mimics Enlight CMF, purpose-built for orthognathic, trauma and reconstruction surgeries involving resection, fibula-based and scapula reconstruction. These planners are designed to be used by clinicians directly. In a demonstration of Enlight CMF, the steps from mirroring a healthy mandible to planning three bone segment placements took under ten minutes – thanks to the planning environment the software solution enables.

“We call Mimics a toolbox,” Beatriz Domínguez González, Market Manager Software in Hospitals and Universities explained. “You open it, and depending on what you need to do that day, you pick your tools. We have also specific anatomy planners where we’ve gone more and more precise. These software tools are very good at doing one specific thing, and makes it as easy as possible so that surgeons themselves can use it.”
The question of who uses the software, engineer or surgeon, turns out to be less “black-and-white” than expected. González noted that while engineers handle the technical segmentation in most hospital settings, many clinicians are capable of using the tools directly because lately advancements made it much more user friendly, particularly within the guided planner interfaces. “It’s more a matter of timing, in some cases. A doctor has limited time availability. So they prefer to have an engineer do most of the segmentation work so they can focus on surgical planning. But they could use it.”
The key advance of 3D planning, she emphasized, is in surface modeling, as opposed to volume rendering. For complex anatomies like congenital hearts, surface-based segmentation is the only approach that achieves the accuracy needed. And the CAD design capabilities layered on top allow teams to move from anatomy to physical guide design within the same platform. “We have that combination of having both,” she said, the diagnostic and the design, in one environment.
Where the gap really is
 If the technology is not the bottleneck, and both Koen Peters and Beatriz González were clear that it no longer is, then what is?
If the technology is not the bottleneck, and both Koen Peters and Beatriz González were clear that it no longer is, then what is?
“I think it’s more on the operational side,” González argues. “It’s embedded into surgical planning. After all, we are bringing new technology, and it takes time.”
The pattern she described is recognizable: one surgeon sees a colleague using technology, asks what it is, tries it, and does not want to go back. Adoption spreads laterally, case by case, specialty by specialty. Technology convinces those who encounter it. The challenge is creating enough encounters.
Reimbursement is the other piece. In the US, a new procedural code for virtual surgical planning is set to take effect in July 2026, a first formal recognition of 3D planning as a distinct clinical activity. Koen was measured in his assessment: “It helps giving 3D planning its place in the system. But the next step is the impact at a reimbursement level, where the actual big financial impact would come in. I don’t think we’re at that stage yet.”
In Europe, the picture is more fragmented. Spain has introduced codes for virtual planning. The Netherlands has a consortium of roughly 20 hospitals working jointly with health authorities. Iceland has had reimbursement for years, though volumes remain small. González described the European effort as a long-term coordination challenge, standardizing evidence across national systems so that what is demonstrated in one country can support the case in another.
“If I achieve reimbursement in Europe,” González said, jokingly, “I can mentally retire. That’s a life mission for me, making sure that every patient who needs this technology can access it, regardless of their postal code.
Koen added a structural observation worth noting: in the US, two specialties (radiology and pediatric cardiology) are the dominant hubs driving adoption, which creates commercial momentum but also concentration. In Europe, adoption is more distributed across specialties, as seen in the clinical applications presented at the forum. This creates diversity of applications, although it makes coordinated advocacy harder.
Turning what seems exceptional into standard of care
 The theme Materialise chose for this year’s forum “Predictability made routine. Care made better” underscores the long road ahead.
The theme Materialise chose for this year’s forum “Predictability made routine. Care made better” underscores the long road ahead.
Aaron James is alive, doing well, returned to normalcy. His case is extraordinary. Today, Materialise and the clinicians at this forum are working toward a future (or may I say, a present) in which the tools that made his care possible are available, systematically, reliably, cost-effectively to any patient who needs them.
As Peters put it: “We are always moving into uncharted territory. That’s part of our DNA. But the value is obvious. The job is to demonstrate it in a structured way, based on data, in the language that decision-makers understand.”
If given the power to change one thing, Peters’ answer was immediate: “Accelerate that demonstration of value, the value that we are obviously providing, in the language of those who make decisions. For the patients.” A wish list of one, but arguably the most important item on any agenda in this space.
For hospitals considering whether to invest in 3D planning capabilities, the message from this forum was consistent: the technology is ready. Workflows exist. The economic case, in operational efficiency alone, is increasingly well-documented. And the clinical outcomes, from orbital reconstruction to mandibular surgery to congenital cardiology, speak for themselves.
The remaining work is still adoption, integration, and the patient accumulation of evidence that turns an impressive case study into a standard of care. That is slower, harder, and less photogenic than a world-first surgery but it is how medicine actually moves forward.
*All images: Courtesy of Materialise